Pheochromocytoma (Paraganglioma)
Clinical
A pheochromocytoma is a paraganglioma located in the adrenal medulla. An inconsistent terminology is in use when describing corresponding extraadrenal neoplasms; some authors refer to them as extraadrenal pheochromocytomas if they are functioning and paragangliomas if nonfunctioning, while others call
all extraadrenal tumors paragangliomas and simply specify the site and functioning status. A paraganglioma originates from chromaffin neural crest tissue that has migrated to form the paraganglionic system. Most are located between the diaphragm and the inferior renal pole, with the most common extraadrenal site being in the organ of Zuckerkandl near the inferior mesenteric artery origin.An occasional one involves the inferior vena cava, urinary bladder, or even the broad ligament.About 10% occur in children, where a familial prevalence is evident and is more likely to be extraadrenal and multicentric. A number of pheochromocytomas have been detected during pregnancy and postpartum. A pheochromocytoma produces an excess of catecholamines, and most of these patients have elevated catecholamine levels. An occasional one is part of a complex tumor; thus it can contain mesenchymal elements. Or, a cortical carcinoma or adenoma exhibits neuroendocrine differentiation. About 10% of pheochromocytomas are malignant. In general, extraadrenal paragangliomas are more malignant and metastasize more readily than their adrenal counterparts. The malignant potential of some is difficult to establish even by histology,
the one definite finding of malignancy being the presence of metastases at sites normally devoid of chromaffin cells.
Although many patients with a pheochromocytoma are hypertensive, overall this condition is a rare cause of hypertension. Pheochromocytoma-
induced hypertension tends to be paroxysmal, but differentiation from other causes of hypertension is difficult. A rare paraganglioma (pheochromocytoma) undergoes spontaneous rupture and extraperitoneal hemorrhage, at times resulting in an acute abdomen (Fig. 16.3). The prevalence of pheochromocytomas is increased in several disorders—neurofibromatosis, von Hippel-Lindau disease, Sturge Weber syndrome, tuberous sclerosis, and multiple endocrine neoplasia (MEN) syndrome. Anatomically, some pheochromocytomacontaining glands are normal in size. Bilateral tumors are more prevalent in both MEN II patients and those with von Hippel-Lindau disease; some of these patients also develop extraadrenal pheochromocytomas. Thus detection of bilateral or familial pheochromocytomas warrants a search for other unsuspected tumors. Of note is that a large minority of these patients with a pheochromocytoma are asymptomatic and have normal blood pressure and normal catecholamine testing. Nevertheless, in patients with von Hippel-Lindau disease and MEN II syndrome, the measurement of plasma normetanephrine and metanephrine achieves a sensitivity and specificity of over 95% in detecting pheochromocytomas (33). Intravenous ionic contrast may precipitate a hypertensive crisis in a patient with a pheochromocytoma. Premedication with an a-adrenergic blocking agent appears prudent prior to intravenous (IV) contrast agent administration to prevent an adrenergic crisis, although the need for such blockage is not well established for nonionic contrast agents.
A pheochromocytoma is a paraganglioma located in the adrenal medulla. An inconsistent terminology is in use when describing corresponding extraadrenal neoplasms; some authors refer to them as extraadrenal pheochromocytomas if they are functioning and paragangliomas if nonfunctioning, while others call
all extraadrenal tumors paragangliomas and simply specify the site and functioning status. A paraganglioma originates from chromaffin neural crest tissue that has migrated to form the paraganglionic system. Most are located between the diaphragm and the inferior renal pole, with the most common extraadrenal site being in the organ of Zuckerkandl near the inferior mesenteric artery origin.An occasional one involves the inferior vena cava, urinary bladder, or even the broad ligament.About 10% occur in children, where a familial prevalence is evident and is more likely to be extraadrenal and multicentric. A number of pheochromocytomas have been detected during pregnancy and postpartum. A pheochromocytoma produces an excess of catecholamines, and most of these patients have elevated catecholamine levels. An occasional one is part of a complex tumor; thus it can contain mesenchymal elements. Or, a cortical carcinoma or adenoma exhibits neuroendocrine differentiation. About 10% of pheochromocytomas are malignant. In general, extraadrenal paragangliomas are more malignant and metastasize more readily than their adrenal counterparts. The malignant potential of some is difficult to establish even by histology,
the one definite finding of malignancy being the presence of metastases at sites normally devoid of chromaffin cells.
Although many patients with a pheochromocytoma are hypertensive, overall this condition is a rare cause of hypertension. Pheochromocytoma-
induced hypertension tends to be paroxysmal, but differentiation from other causes of hypertension is difficult. A rare paraganglioma (pheochromocytoma) undergoes spontaneous rupture and extraperitoneal hemorrhage, at times resulting in an acute abdomen (Fig. 16.3). The prevalence of pheochromocytomas is increased in several disorders—neurofibromatosis, von Hippel-Lindau disease, Sturge Weber syndrome, tuberous sclerosis, and multiple endocrine neoplasia (MEN) syndrome. Anatomically, some pheochromocytomacontaining glands are normal in size. Bilateral tumors are more prevalent in both MEN II patients and those with von Hippel-Lindau disease; some of these patients also develop extraadrenal pheochromocytomas. Thus detection of bilateral or familial pheochromocytomas warrants a search for other unsuspected tumors. Of note is that a large minority of these patients with a pheochromocytoma are asymptomatic and have normal blood pressure and normal catecholamine testing. Nevertheless, in patients with von Hippel-Lindau disease and MEN II syndrome, the measurement of plasma normetanephrine and metanephrine achieves a sensitivity and specificity of over 95% in detecting pheochromocytomas (33). Intravenous ionic contrast may precipitate a hypertensive crisis in a patient with a pheochromocytoma. Premedication with an a-adrenergic blocking agent appears prudent prior to intravenous (IV) contrast agent administration to prevent an adrenergic crisis, although the need for such blockage is not well established for nonionic contrast agents.
Spontaneous rupture of paraganglioma. A: Contrast-enhanced CT shows a retroperitoneal tumor with peripheral enhancement (arrows). B: A more caudad scan identifies left para-aortic fluid and infiltrate, mimicking a ruptured aortic aneurysm.
Imaging
A review of 282 patients who underwent
pheochromocytoma resection in France
between 1980 and 1991, found unilateral tumors
in 67%, bilateral ones in 19%, and extraadrenal
in 14% (34); the sensitivities of imaging in
detecting these tumors were 89% for CT, 98% for
MRI, and 81% for I-131-MIBG scintigraphy.
If imaging reveals no adrenal tumor in a
patient suspected of a pheochromocytoma,
imaging of other extraadrenal sites, including
bladder, is necessary. Scintigraphy with I-131-
MIBG is useful to detect extraadrenal and bilateral
tumors.
Most pheochromocytomas are readily
imaged by CT, US, and MRI (Fig. 16.4), yet the
clinical and imaging findings are not always
straightforward, even in a setting of elevated
catecholamines. Intrinsically solid tumors,
necrosis, and hemorrhage result in a cystic
appearance and, as a result, they have a variable
imaging appearances. It is with cystic tumors
that the differential diagnosis between cystic
pheochromocytomas, necrotic carcinomas,
and metastases becomes problematic. An aid to
diagnosis is that aside from necrotic regions,
these are very hypervascular tumors and
postcontrast CT shows marked contrast
enhancement.
Imaging
A review of 282 patients who underwent
pheochromocytoma resection in France
between 1980 and 1991, found unilateral tumors
in 67%, bilateral ones in 19%, and extraadrenal
in 14% (34); the sensitivities of imaging in
detecting these tumors were 89% for CT, 98% for
MRI, and 81% for I-131-MIBG scintigraphy.
If imaging reveals no adrenal tumor in a
patient suspected of a pheochromocytoma,
imaging of other extraadrenal sites, including
bladder, is necessary. Scintigraphy with I-131-
MIBG is useful to detect extraadrenal and bilateral
tumors.
Most pheochromocytomas are readily
imaged by CT, US, and MRI (Fig. 16.4), yet the
clinical and imaging findings are not always
straightforward, even in a setting of elevated
catecholamines. Intrinsically solid tumors,
necrosis, and hemorrhage result in a cystic
appearance and, as a result, they have a variable
imaging appearances. It is with cystic tumors
that the differential diagnosis between cystic
pheochromocytomas, necrotic carcinomas,
and metastases becomes problematic. An aid to
diagnosis is that aside from necrotic regions,
these are very hypervascular tumors and
postcontrast CT shows marked contrast
enhancement.
A: Left adrenal pheochromocytoma. Computed tomography reveals a large, homogeneous, poorly enhancing tumor
(arrows) displacing the left kidney. B: Right adrenal pheochromocytoma. Computed tomography identifies a large, homogeneous,
poorly enhancing tumor (arrows).
(arrows) displacing the left kidney. B: Right adrenal pheochromocytoma. Computed tomography identifies a large, homogeneous,
poorly enhancing tumor (arrows).
Another source for confusion is that a minority
of adrenal pheochromocytomas contain
sufficient microscopic fat to result in a CT attenuation
of <10HU and thus mimic an adenoma
(35); after contrast enhancement some of these
hypodense tumors also reveal >60% contrast
washout on 10-min images, similar to
adenomas.
Some contain linear or laminated calcifications
(Fig. 16.5).Aside from several anecdotal
reports, pheochromocytomas do not contain
sufficient lipid to influence their imaging
appearance.
(Fig. 16.5).Aside from several anecdotal
reports, pheochromocytomas do not contain
sufficient lipid to influence their imaging
appearance.
Left adrenal pheochromocytoma with liver metastasis.
A: Precontrast CT reveals a rim-calcified tumor in the pancreatic
tail (arrow). B,C: Two contrast-enhanced CT images show
lack of tumor enhancement.A mottled appearance in the liver was
due to extensive metastases.
A: Precontrast CT reveals a rim-calcified tumor in the pancreatic
tail (arrow). B,C: Two contrast-enhanced CT images show
lack of tumor enhancement.A mottled appearance in the liver was
due to extensive metastases.
Nonnecrotic pheochromocytomas tend to
be hypointense-to-isointense to liver on T1-
and hyperintense on T2-weighted images. Their
lack of fat reflects their hyperintense T2-
weighted fat-suppressed appearance. They tend
to exhibit progressive enhancement postcontrast
MR. Nevertheless, a sufficient number
of pheochromocytomas have an atypical low
signal intensity on T2-weighted images and
not all hyperintense adrenal tumors represent
pheochromocytomas, so that reliance on a
hyperintense T2-weighted appearance results in
a low sensitivity in diagnosing a pheochromocytoma
(Fig. 16.6).
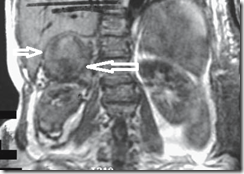
Bleeding pheochromocytoma (arrows) identified
on T1-weighted coronal image. Hypointensity surrounding
the right kidney represents a perinephric and paranephric
hematoma.
on T1-weighted coronal image. Hypointensity surrounding
the right kidney represents a perinephric and paranephric
hematoma.
Scintigraphy with I-123-MIBG achieves an
80% to 90% detection rate for these tumors;
MIBG SPECT sensitivity approaches 100%.
This tracer accumulates in adrenergic tissue
throughout the body, including metastases.
Optimal scan timing is variable, with scans
often obtained 24 to 48 hours postinjection. An
occasional metastasis is detected only on earlier
scans. Indium-111 pentetreotide scintigraphy
appears to have similar detection ability as
I-123-MIBG, but it has not been studied as
extensively.
2-[18F]-fluoro-deoxy-D-glucose PET detected
tumors in 76% of patients with
pheochromocytomas, with most benign, malignant,
and metastatic foci avidly concentrating
FDG (36); in fact, several pheochromocytomas
not accumulating MIBG showed intense FDG
uptake, although MIBG images tended to be as
good or better for tumors concentrating both
agents. A majority of pheochromocytomas also
reveal uptake during (11C)-hydroxyephedrine-
PET scanning (37).
Therapy
The treatment of choice for most pheochromocytomas
is surgical resection, although an
occasional one is treated by catecholamine
pharmacotherapy. Resection consists of either
adrenalectomy or adrenal-sparing surgery, with
a laparoscopic approach commonly employed.
A pheochromocytoma has been treated with
percutaneous radiofrequency ablation (23).



Post a Comment for "Pheochromocytoma (Paraganglioma)"